
The same Stan Meyer fuel cell today.

Stanley Meyer “fuel cell” producing hydroxy gas (Grove City Record, Oct. 25, 1984).
Techniques for using electrolysis to break apart water molecules into a mixture of hydrogen and oxygen had been known centuries before Michael Faraday did his quantitative electrolysis experiments in the 1830s. Today the mixture of gases produced by electrolysis of water is commonly termed “Hydroxy Gas” or Brown’s gas (named after Yul Brown who researched and promoted this technology for many years). A detailed history of hydroxy gas can be found in Robey (2006).
See also: The Orion Project Visits Stan Meyer’s Technology

Stan Meyer’s ‘Water Powered Car’
as seen in January, 2008.
Many researchers have developed techniques for producing this gas in order to add it into the fuel stream of a fossil fuel burning engine, where it has been shown to increase the efficiency of the engine and reduce pollutants by a claimed MPG percent increase of between 10 to over 50%. One of the many problems with this technology is that a lot of electrical energy has to be used to produce the gas, so that burning the gas alone to power a generator to produce power to produce the gas just does not work. There are just too many inefficiencies in the loop.
However, in recent years a number of different inventors have developed technologies that produce hydroxy gas in quantities much greater than would be predicted using standard “brute force” electrolysis. These techniques typically include a combination of multiple high frequency pulsed voltages applied to specially designed electrolysis cells that resonate the water molecule, basically causing it to “shake” apart with a much reduced amount of electrical power per unit gas produced.
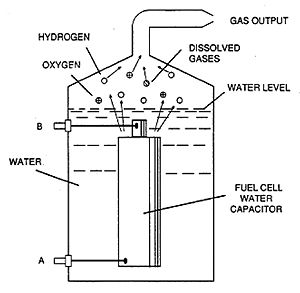
Drawing from Stanley A. Meyer
US Patent 4,936,961, June 26, 1990.
Some of these techniques are described in research papers by Meyer (1997) and Griffin (1993). We are presently working with several of these research groups with the intent of bringing them together to optimize these technologies in order to be able to close the loop and produce a 5-10Kw generator running only on water.